Cause Analysis and Solutions for Ethylene Pipeline Elbow Corrosion
Abstract: During a scheduled inspection at a petrochemical plant, pitting corrosion was detected in the base material of a TP304 ethylene pipeline elbow. To determine the cause and develop effective countermeasures, a comprehensive analysis was conducted, including evaluation of the base material’s chemical composition, macroscopic defect morphology, corrosion product composition, metallographic structure, hardness, and pressure resistance. Since TP304 stainless steel is widely used in petrochemical engineering, understanding its corrosion mechanisms and implementing preventive measures is crucial for ensuring the long-term safe operation of petrochemical facilities. The ethylene pipeline at the petrochemical plant was completed in September 2007 and commissioned in October 2008. During a scheduled inspection in May 2013, localized corrosion and wall thinning were observed. The affected section was located on a horizontal segment between two pipe racks (Figure 1). The ethylene pipeline, designated 300-P-0501007-A21L19-R, is made of TP304 stainless steel, with its specifications provided in Table 1.
1. Investigation of Wall Thinning and Corrosion in Ethylene Pipeline Elbows
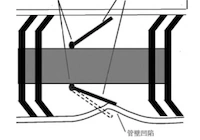
During the equipment overhaul in May 2013, a routine inspection of the ethylene pipeline was carried out, including radiographic (RT) spot checks on several welds. The relevant images are shown in Figures 1–5. Several welds exhibited areas of abnormally dark contrast on the RT films, all of which were located between two pipe galleries. A typical RT defect image is shown in Figure 2. According to NB/T 47013.2–2015, Nondestructive Testing of Pressure Equipment – Radiographic Testing, such abnormally dark regions generally indicate a reduction in pipe wall thickness, which was later confirmed by ultrasonic thickness measurements in the affected areas. To assess the pipe’s actual condition, the construction company sampled an elbow located beneath the pipe galleries (Figure 3). Upon cutting, visible corrosion marks (Figure 4) and corrosion pits (Figure 5) were observed on the elbow’s base metal.

Figure 1 Horizontal ethylene pipeline between two pipe galleries
Table 1 Ethylene pipeline process parameters
|
Nominal diameter / mm |
Wall thickness / mm |
Length / m |
Design pressure / MPa |
Operating pressure / MPa |
Design temperature / °C |
Operating temperature / °C |
Medium |
Material |
Pipeline grade |
|
300 |
5 |
578 |
1.1 |
0.25 |
−101 |
65 |
Ethylene |
TP304 |
GC2 |

Figure 2 RT film showing abnormal dark regions

Figure 3 Sampled elbow section under pipe gallery

Figure 4 Corrosion marks on the elbow base metal
Figure 5 Corrosion pits at the weld edge of the elbow base metal
Following the discovery, the construction unit immediately brought together the relevant departments for joint analysis and discussion. The pipeline operated continuously from its commissioning in October 2008 until it was shut down for maintenance in May 2013. During this maintenance period, routine inspections revealed abnormal dark areas on the radiographic films of several weld joints. Severe pitting corrosion was concentrated on the elbows beneath the portal-type pipe gallery, with the maximum measured wall thinning reaching 2.0 mm, equivalent to 40% of the nominal 5 mm wall thickness. The initial diagnosis indicated chloride ion–induced pitting corrosion; however, because ethylene does not contain chloride ions, the source of chloride contamination in the pipeline remained unclear. In response, the meeting directed the Materials Department to urgently procure TP304 material for repairs, while the Equipment Department reviewed installation records to verify the base material specifications, welding procedures, and nondestructive testing documentation. Additionally, a failure analysis was commissioned on the corroded pipe sections, and this article presents and discusses the findings.
2. Chemical Composition of Sampled Pipe Sections
Based on the standard chemical composition of TP304 stainless steel in Appendix A.2 of SH/T 3523-2009, ‘Petrochemical Chromium-Nickel Stainless Steel, Iron-Nickel Alloy, and Nickel Alloy Welding Specification,’ chemical composition analyses were conducted at six points on the parent material of the straight pipe, the welds, and the elbow, with results summarized in Table 2. The analysis indicates that the chemical compositions of the straight pipe, weld metal, and elbow base metal all meet the standard requirements for TP304 stainless steel.
Table 2 Chemical Composition of Base Metal (mass %)
|
No. |
Sampling Location |
C |
Si |
Mn |
P |
S |
Cr |
Ni |
Fe |
|
Standard (SH/T 3523-2009) |
— |
≤ 0.08 |
≤ 0.75 |
≤ 2.0 |
≤ 0.04 |
≤ 0.03 |
18–20 |
8–11 |
Balance |
|
1 |
Straight pipe (left) |
0.064 0 |
0.490 3 |
1.602 |
0.023 4 |
0.003 7 |
18.29 |
8.29 |
Balance |
|
2 |
Straight pipe (right) |
0.062 1 |
0.325 2 |
1.432 |
0.038 4 |
0.003 5 |
19.23 |
9.39 |
Balance |
|
3 |
Elbow (upper) |
0.046 5 |
0.345 6 |
1.772 |
0.039 1 |
0.004 6 |
18.27 |
8.03 |
Balance |
|
4 |
Elbow (lower) |
0.048 0 |
0.384 7 |
1.571 |
0.006 6 |
0.002 0 |
18.20 |
8.11 |
Balance |
|
5 |
Weld 1 |
0.059 5 |
0.417 2 |
1.731 |
0.040 0 |
0.005 1 |
19.88 |
9.19 |
Balance |
|
6 |
Weld 2 |
0.044 1 |
0.509 1 |
1.522 |
0.039 2 |
0.007 7 |
18.43 |
8.00 |
Balance |
3. Macroscopic Defect Morphology and Corrosion Product Analysis
3.1 Macroscopic Defect Morphology
Two corroded pits on the elbow were cleaned and cross-sectioned for examination, with the results shown in Figure 6. As illustrated, the corrosion defects are located in the base metal, with the pit edges extending both along the surface and into the wall thickness of the pipe. According to ASTM G46, ‘Standard Guide for Examination and Evaluation of Pitting Corrosion,’ and GB/T 18590-2001, ‘Corrosion of Metals and Alloys – Pitting Corrosion Assessment Methods,’ Section 3.1.4.1, the typical cross-sectional shapes of pitting corrosion are shown in Figure 7. The macroscopic morphology of the examined corrosion pits matches these characteristic profiles, representing a typical case of localized pitting corrosion. The shape and extent of these pits are influenced by factors such as corrosion potential, medium composition, metal microstructure, and temperature, with chloride (Cl⁻) ions being a key contributor to pitting in austenitic stainless steels.

Figure 6 Cross-sectional morphology of pitting in the sampled area
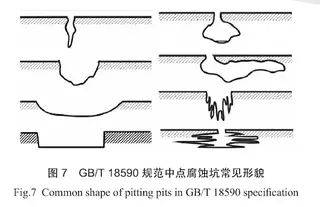
Figure 7 Common pitting morphologies from GB/T 18590
3.2 Energy Dispersive Spectroscopy (EDS) Analysis of Corrosion Products
In accordance with Section 3.1.1 of GB/T 18590-2001, ‘Corrosion of Metals and Alloys — Pitting Corrosion Assessment Methods,’ which states that the composition of corrosion products provides valuable insights into the cause of corrosion, energy dispersive spectroscopy (EDS) was performed on the corrosion products within the elbow pits. The analysis revealed the presence of chlorine (Cl) and sulfur (S) in the corrosion products, with detailed results presented in Table 3.
Table 3 Energy Dispersive Spectroscopy (EDS) Results of Corrosion Products
|
Element |
C |
O |
Al |
Si |
S |
Cl |
Ca |
Cr |
Mn |
Fe |
Ni |
|
Mass/% |
1.99 |
13.98 |
0.14 |
0.84 |
0.54 |
1.90 |
0.13 |
0.34 |
17.68 |
1.57 |
55.93 |
As shown in Table 3, chlorine (Cl) constitutes 1.90% of the corrosion products, ranking as the sixth most abundant element. Chloride ions tend to accumulate at the bottom of corrosion pits, directly promoting pitting and stress corrosion cracking in TP304 stainless steel. The sulfur content in the corrosion products reaches 0.54%, far exceeding the maximum allowable limit for TP304 base material (≤ 0.03%), and the presence of sulfur ions further accelerates both the initiation and propagation of pitting corrosion. Based on the macroscopic pit morphology and the chemical composition of the corrosion products, it can be concluded that the defects observed in the sampled elbow were caused by chloride-induced corrosion of the stainless steel, resulting in rapid material degradation and significant wall thinning.
4. Metallographic Structure
To ensure representative results, metallographic analysis was conducted on the sampled pipe section, focusing separately on the base material, the corrosion pit at the weld edge, and the corrosion pit within the base metal.
4.1 Metallographic Structure of the Elbow Base Material
Figure 8 shows the sampling locations for metallographic analysis of the elbow base material, while Figure 9 presents the metallographic structures of its inner and outer surfaces at 200× magnification. The microstructure indicates that the base metal received proper solution treatment, producing a uniform and stable austenitic structure, with no evidence of carbide precipitation or sensitization.

Figure 8 Schematic diagram showing sampling locations for metallographic analysis of the elbow base material
Figure 9 Metallographic structure of the inner and outer surfaces of the elbow base material
(a) Inner surface, 200× (b) Outer surface, 200×
4.2 Metallographic Structure of Corrosion Pit at the Weld Edge
Figure 10 shows the sampling locations of corrosion pits near the weld edge, and Figure 11 presents the corresponding metallographic profiles.

Figure 10 Sampling location of corrosion pits at the weld edge
Figure 11 Metallographic profiles of pitting corrosion at the weld edge
(a) Micro-pitting parallel to the surface, 100× (b) Magnified view of (a), 500×
(c) Microcracks extending from the pit bottom (d) Magnified view of (c), 200×
As shown in Figure 11, the pitting corrosion penetrates the wall thickness, with microcracks developing along both the sides and the bottom of the pits. The lateral cracks display characteristics of transgranular stress corrosion cracking (TGSCC), while additional microcracks are observed at the bottom of the pits. These cracks are primarily caused by stress concentration at the pit bottoms and residual stresses in the weld region, which together facilitate stress corrosion cracking. Additionally, sensitization at the weld, resulting from carbide precipitation along the grain boundaries, promotes intergranular crack propagation and further exacerbates the corrosion damage.
4.3 Metallographic Structure of Base Metal Pit
Figure 12 presents a schematic diagram of the sampling locations used for metallographic examination of corrosion pits in the base metal. Surface A corresponds to the pipe’s axial surface, with its metallographic structure shown in Figure 13, while Surface B corresponds to the circumferential surface, shown in Figure 14.

Figure 12 Schematic diagram of metallographic sampling of corrosion pits in base material
Figure 13 Axial metallographic view of corrosion pits
(a) Axial view of corrosion pit 50×
(b) Magnified view of (a) at 200×
Figure 13 presents the axial metallographic view of the corrosion pit, showing the pit bottom morphology.
Figure 14 Circumferential metallography of corrosion pits
(a) Circumferential view of corrosion pit 50× (b) Magnified view of (a) at 100×
(c) Magnified view of (b) at 200× (d) Magnified view of (a) at 200×
Figure 14 shows the circumferential metallographic images of the corrosion pit, where intergranular corrosion is evident and transgranular microcracks at the pit bottom indicate stress corrosion cracking. The metallographic results reveal distinct corrosion characteristics depending on orientation: in the axial direction, intergranular corrosion predominates along grain boundaries, while in the circumferential direction, transgranular microcracks develop across grains, indicating a combined effect of localized corrosion and stress corrosion cracking.
6. Failure Cause Analysis
(1) Defect location and material verification: Since the detected defects in the ethylene pipeline are confined to the base metal, and both the hardness and chemical composition of the steel meet standard requirements, the welding process and quality can be ruled out as contributing factors.
(2) Corrosion mechanism: Based on the analyses of chemical composition, hardness, metallographic structure, and corrosion products, no abnormalities were detected in the manufacturing quality of the elbow. The corrosion defect is attributed to chloride ion–induced pitting of the TP304 stainless steel within the pipeline, with sulfur ions accelerating the corrosion and promoting the progression of pitting into both longitudinal and transverse linear corrosion.
(3) Pressure test and operational history: According to installation records, the ethylene pipeline underwent sectional hydrostatic testing in September 2007 using pure water supplied by the construction contractor, although no water quality report was retained. After passing the pressure test, the water was discharged, and the golden joint was subsequently welded in place. The golden joint was located midway between two pipe corridors, approximately 200 meters from each side. At that time, other auxiliary systems—such as nitrogen and air systems—were incomplete, preventing pipeline purging. After the golden joint passed inspection, the system remained idle until late September 2008, when the airtightness test and full-system purging were completed, and the process was commissioned. Within the corridor area, the ethylene pipelines consisted entirely of straight runs without valves or drain points. After the hydrostatic test, although the water was discharged, residual water inevitably accumulated in the horizontal sections between the corridors. During the one-year idle period, residual chloride and sulfur ions in the stagnant water concentrated in low-lying areas, leading to pitting corrosion of the TP304 stainless steel. As a result, corrosion defects were concentrated in the low-lying horizontal sections between the two corridors, with the elbow beneath the corridor—being at the lowest elevation—experiencing the most severe corrosion.
(4) Corrosion morphology observations:
Further analysis of the corroded pipe section (Figures 15 and 16) supports these findings. In Figure 15, a prominent rust band is visible along the upper inner wall of the pipe. This band formed as residual water from the hydrostatic test evaporated, leading to localized condensation and droplet accumulation on the upper surface. The high chloride concentration within these droplets promoted strong localized corrosion, resulting in the formation of rust. In Figure 16, corrosion traces spread laterally along the pipe’s circumference and then extend downward under the influence of gravity, forming distinct vertical corrosion streaks. Additionally, due to the capillary effect along the pipe wall surface, moisture within the corrosion pits migrated upward, resulting in the upward extension of the corrosion marks.

Figure 15 Rust bands on the inner wall of the pipe
Figure 16 Corrosion pit morphology on the inner wall of the pipe
7. Conclusion and Countermeasures
7.1 Conclusion
The pipeline defect was identified as pitting corrosion failure of TP304 stainless steel, caused by chloride ions originating from residual water left in the pipeline after the hydrostatic test conducted in September 2007. During the idle period, residual ions accumulated in the low-lying areas of the pipeline, leading over time to localized pitting and thinning of the stainless steel wall.
7.2 Countermeasures
(1) To ensure timely resumption of production, replace all pipe sections with abnormal darkening observed during inspection. After the new welds successfully pass nondestructive testing, proceed with pressure testing under the supervision of the production department.
(2) The chloride ion concentration in the hydrostatic test water shall be strictly controlled to not exceed 2.5 × 10⁻⁶. Upon completion of the pressure test, the water shall be immediately drained, and the pipeline thoroughly dried using nitrogen. The process medium should then be introduced as soon as possible to minimize idle time, thereby diluting and removing any residual chloride ions and preventing their accumulation in low-lying areas.
(3) After system commissioning, the operations department shall enhance monitoring and implement specific protective measures to facilitate early detection of corrosion, ensuring stable and safe operation.
(4) The materials department shall ensure the timely preparation of all necessary replacement materials and coordinate the complete pipeline replacement during the next scheduled maintenance period.
(5) The scheduling of construction, pressure testing, purging, and commissioning in the ethylene pipeline maintenance procedures shall be optimized. The pipeline must remain dry during construction, and no residual water should be left after pressure testing. Pressure testing, purging, and commissioning should be performed as continuously as possible to minimize idle time and prevent the recurrence of similar corrosion failures.
Send your message to this supplier
Related Articles from the Supplier
Related Articles from China Manufacturers
Related Products Mentioned in the Article
Supplier Website
Source: https://www.landeepipefitting.com/cause-analysis-and-solutions-for-ethylene-pipeline-elbow-corrosion.html