σ Phase Formation and Microstructure in Austenitic Heat-Resistant Steel Pipe Joints
Abstract: This study investigates the formation of the σ phase in austenitic heat-resistant steel pipe joints used in power plant boilers. Results show that different morphologies of the σ phase precipitate in austenitic steel joints after high-temperature aging. The formation of the σ phase is mainly controlled by chemical composition and temperature. Among these factors, composition exerts the most critical and complex influence. The amount of σ phase increases with aging temperature and time, while excessive heat input further accelerates its precipitation. During high-temperature creep, a network-like σ phase forms in the coarse-grained heat-affected zone (HAZ), significantly reducing the creep rupture strength of the joint. Effective control of σ phase precipitation requires optimizing the weld metal composition, along with appropriate heat input and process parameters, to ensure structural stability and extend service life.
Introduction
Superheater and reheater tubes in ultra-supercritical power plant boilers operate at temperatures up to 650 °C and beyond, necessitating the use of austenitic or advanced austenitic heat-resistant steel pipes. This service temperature falls within the precipitation range of intermetallic phases such as the σ phase. The σ phase is an intermetallic compound with a complex tetragonal lattice, enriched in Fe and Cr, and characterized by variable composition. It can form in over 50 transition alloys across a wide temperature range, with the most rapid precipitation occurring between 600 °C and 900 °C. In Cr–Ni austenitic steels, a chromium content of 17–20% promotes σ phase formation, whereas higher nickel content suppresses it. In general, all ferrite-forming elements—such as Cr, Nb, Ti, Mo, W, and Si—promote σ phase precipitation. The σ phase can precipitate either directly from austenite or from ferrite. Because of differences in alloying element diffusion rates, σ phase formation in austenite is roughly 100 times slower than in ferrite. However, the presence of δ-ferrite in austenitic alloys significantly accelerates its precipitation. The σ phase is hard and brittle, with a hardness of 900–1000 HV. Even trace amounts in austenitic steels or welded joints can drastically impair toughness and ductility. In superheater tubes, elbows, and welded joints exposed to prolonged high-temperature service, σ phase precipitation can markedly reduce creep strength or cause dish-shaped cracking, ultimately resulting in premature failure of critical components. Although many studies have investigated phase formation in homogeneous stainless steels and its impact on performance, relatively few have addressed the behavior of the σ phase in heat-resistant steel tubes and joints for power plant boilers. Comprehensive studies on the conditions and mechanisms of σ phase formation in these joints remain limited. This paper analyzes the conditions of σ phase precipitation, its influencing factors, and its impact on fracture behavior in welded joints, while also discussing the principles for controlling its formation. This research offers both theoretical guidance and practical reference for evaluating the safety of austenitic heat-resistant steel joints, optimizing their microstructure and properties, and extending their service life.
1. Microstructure and Formation Conditions of the σ Phase
1.1 Microstructure of Phases
The optical microstructures of phases in austenitic stainless steels under various service conditions are shown in Figure 1. In rolled AISI 316 steel (Figure 1a), the σ phase precipitates within δ-ferrite grains, exhibiting a characteristic “cauliflower-like” morphology. Due to rolling, precipitation occurs along a fixed crystallographic orientation. In AISI stainless steel with a 50% reduction (Figure 1b), the σ phase appears as finely dispersed, spherical precipitates. Optical metallographs of HR3C steel welded joints before and after aging at 650 °C are shown in Figure 2, and a comparison of the microstructures is summarized in Table 1. Before aging, the HR3C base metal comprises equiaxed austenite grains containing a small number of Laves-phase particles within the grains, along with a few dispersed precipitates at the grain boundaries and inside the grains (see Figure 2a). In the fusion zone, the austenite displays dendritic and cellular morphologies, nucleating along the fusion line of the base material and growing in the direction opposite to heat dissipation (see Figure 2b). In the weld metal, a small number of granular precipitates are present along the columnar or cellular grain boundaries (see Figure 2c). After aging at 650 °C for 7000 hours, continuous, network-like precipitates form along the grain boundaries of the base metal, with localized spheroidization and coarsening. Within the grains, precipitates appear in dense spherical or blocky forms (see Figure 2d). In the fusion zone, precipitates solidify into a network pattern, while those along subgrain boundaries form needle-like or short rod-shaped clusters, exhibiting higher density than in the base metal (see Figure 2e). In the weld metal, numerous granular precipitates are distributed both within the austenite grains and along their boundaries (see Figure 2f). Figure 3 shows TEM images of HR3C steel welded joints aged at 650 °C for 7000 hours. The continuous, chain-like precipitates along the grain boundaries are primarily M₂₃C₆ carbides (see Figure 3a). Large spherical and blocky precipitates are observed within the grains, along with a minor fraction of Nb(C,N) (see Figure 3b). Selected-area electron diffraction (SAED) and energy-dispersive spectroscopy (EDS) analyses confirm that the long, needle- or rod-shaped precipitates near the grain boundaries are Cr-rich σ-phase (see Figure 3c).
Weld metals of three austenitic stainless steel electrodes in the as-welded state and after heat treatment (840 °C normalizing + 840 °C quenching + 520 °C tempering):

a. As-rolled AISI 316 stainless steel b. AISI stainless steel with 50% reduction
Figure 1. Microstructure of austenitic stainless steel phases under different service conditions
Joint Specimen Condition: Weld Zone, Fusion Zone, Base Metal Zone
As-Welded State:
In the weld zone, small amounts of granular precipitates are present along columnar or cellular grain boundaries (see Figure 2c). In the fusion zone, austenite displays dendritic and cellular morphologies, nucleating along the fusion line of the base metal and growing in the direction opposite to heat dissipation (see Figure 2b). The base metal is composed of equiaxed austenite grains containing a few twins, with precipitates distributed both along the grain boundaries and within the grains (see Figure 2a).
After aging at 650 °C for 7000 h
Numerous granular precipitates develop both within the austenite grains and along the grain boundaries of the weld metal (see Figure 2f). In the fusion zone, precipitates formed during solidification crystallize into a network structure, while subgrain-boundary precipitates appear as clusters of needles or short rods, with a density higher than that of the base metal (see Figure 2e). In the base metal, continuous network-like precipitates develop along the grain boundaries, with localized spheroidization. Intragranular precipitates are present as densely packed spherical or blocky particles (see Figure 2d). A comparison of these microstructures is presented in Table 2. In the as-welded state, all three electrode samples displayed an austenite + δ-ferrite structure, although the δ-ferrite content and morphology differed among them. After heat treatment, all three samples exhibited a microstructure composed of austenite + δ-ferrite + M₂₃C₆ carbide + G-phase, with numerous fine brown precipitates appearing within the δ-ferrite grains and along the austenite–ferrite grain boundaries. High-magnification optical microscopy of hematite-stained specimens under a Neophot-2 metallograph, combined with microhardness testing, confirmed the presence of σ-phase, which is rarely observed within austenite grains. Among the three electrodes, sample B exhibited the highest σ-phase content, whereas samples A and C contained lower amounts. Table 3 summarizes the morphology of precipitates under various processing conditions. The σ-phase morphology and precipitation sites are highly dependent on the specimen’s state and the applied heat treatment. Examples 1 and 2 represent as-rolled AISI 316 stainless steel, in which σ-phase precipitation occurs during the rolling process. In Figure 1a, shown at higher magnification than Figure 1b, the morphology and distribution of the σ-phase are more clearly visible. Example 3 represents an HR3C stainless steel welded joint aged at 650 °C for 7000 hours. High-resolution TEM analysis, coupled with electron diffraction and EDS, confirmed that the long, needle- or rod-shaped Cr-rich precipitates near the grain boundaries are σ-phase. The chemical compositions of AISI 316 and HR3C stainless steels are listed in Table 4. In Example 4, hematite-etched specimens of the deposited weld metal were examined under a Neophot-2 metallograph at high magnification. Microhardness testing confirmed that the numerous brown precipitate clusters within δ-ferrite grains and at austenite–ferrite boundaries are σ-phase.
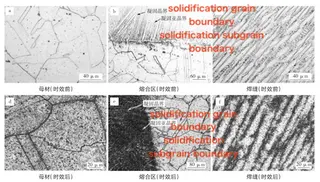
Figure 2 Optical microstructures of HR3C steel welded joints before and after aging at 650°C
a. Base metal (before aging) b. Fusion zone (before aging) c. Weld metal (before aging) d. Base metal (after aging) e. Fusion zone (after aging) f. Weld metal (after aging)

Figure 3 TEM images of HR3C steel welded joints aged at 650°C for 7000 h
a. M₂₃C₆ at grain boundaries in the weld b. M₂₃C₆ and Nb(C,N) within the weld grains c. σ-phase near the grain boundaries of the weld
Table 2. Microstructure of deposited metal of austenitic stainless steel electrodes in the as-welded and heat-treated states
|
Electrode No. |
Alloy System |
Cr/Ni Equivalent Ratio |
δ-Ferrite Content (%) |
As-welded Microstructure |
Post-weld Heat-treated Microstructure |
|
A |
Cr25-Ni13 |
1.77 |
6–7 |
γ + δ-ferrite |
γ + δ-ferrite + M₂₃C₆ + G-phase |
|
B |
Cr30-Ni12 |
1.96 |
23–25 |
γ + δ-ferrite |
γ + δ-ferrite + M₂₃C₆ + G-phase |
|
C |
Cr20-Ni10-Mn6 |
1.46 |
6.5–7.5 |
γ + δ-ferrite |
γ + δ-ferrite + M₂₃C₆ + G-phase |
Table 3. Phase morphology of precipitates under different process conditions
|
Example |
G-phase Morphology |
Precipitation Location |
Sample State |
Sample Material |
|
1 |
Clustered “cauliflower-like” |
Inside δ-ferrite grains |
Rolled state |
AISI 316 stainless steel |
|
2 |
Dispersed spherical |
At austenite grain boundaries or inside grains |
50% rolled |
AISI 316 stainless steel |
|
3 |
Long needle-/rod-shaped |
Near grain boundaries in weld |
Aged 650°C × 7000 h |
HR3C stainless steel |
|
4 |
Brown particulate clusters |
Inside δ-ferrite grains and at γ/δ boundaries |
Heat-treated (840°C normalize + 840°C quench + 520°C temper) |
Austenitic stainless steel electrode deposited metal |
Table 4. Chemical composition of two austenitic stainless steels (wt.%)
|
Steel Type |
C |
Si |
Mn |
P |
S |
Cr |
Ni |
Mo |
Nb |
N |
|
316 |
0.08 |
1.0 |
2.0 |
0.045 |
0.03 |
16.0–18.0 |
10.0–14.0 |
2.0–3.0 |
— |
— |
|
HR3C |
0.10 |
1.5 |
2.0 |
0.030 |
0.03 |
23.0–27.0 |
17.0–23.0 |
— |
0.20–0.60 |
0.15–0.35 |
1.2 Phase Formation Conditions
As shown in Tables 1–3, σ-phase precipitation occurs only in welded joint specimens following post-weld heat treatment. In contrast, M₂₃C₆ carbide precipitation is unavoidable in the as-welded specimens. Although carbides can be detected at temperatures above 500 °C, essentially no σ-phase forms under these conditions. Two fundamental conditions must be met for σ-phase formation: chemical composition and temperature.
Composition: The σ-phase typically precipitates only when the chromium content exceeds approximately 16%. Furthermore, due to chromium’s high diffusivity, σ-phase forms more rapidly in δ-ferrite than in γ-austenite. This compositional requirement is considered a necessary condition for σ-phase formation.
Temperature: The 600–900 °C range is considered the critical window for σ-phase precipitation. Maintaining the temperature within this range is thus a sufficient condition. Both compositional and thermal criteria must be satisfied simultaneously for σ-phase formation.
The rate of σ-phase formation remains a subject of debate, with several perspectives proposed:
- One perspective holds that within the 600–800 °C range, σ-phase forms very slowly, requiring tens to hundreds of hours to precipitate.
- Equilibrium is achieved only after the prior precipitation of χ-phase, particularly in ternary alloy systems.
- Another perspective holds that in duplex stainless steels, σ-phase forms rapidly, with G-phase precipitating within 0.5–2 hours of exposure. The transformation rate peaks at approximately 810 °C, completing in as little as 30 minutes.
- A third perspective maintains that the σ-phase transformation rate increases steadily with rising temperature.
Vitek et al. proposed a distinctive explanation for σ-phase transformation kinetics, suggesting that in most χ-to-σ transformations, the nucleation of σ-phase is the rate-limiting step. Once nucleated, σ-phase grows relatively easily. During nucleation, the crystallographic rearrangement is the key factor, whereas chromium diffusion plays only a secondary role. Thus, diffusion is not the primary factor controlling the transformation rate. Under typical welding thermal cycles, the weld metal remains at 600–800 °C for a relatively short duration, which is insufficient for substantial σ-phase formation. As a result, σ-phase does not form in as-welded specimens.
Send your message to this supplier
Related Articles from the Supplier
Related Articles from China Manufacturers
Related Products Mentioned in the Article
Supplier Website
Source: https://www.landeepipefitting.com/s-phase-formation-and-microstructure-in-austenitic-heat-resistant-steel-pipe-joints.html


